 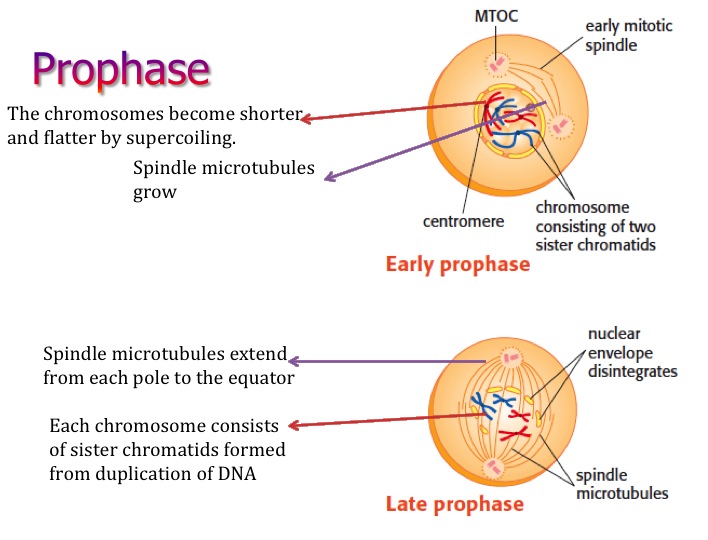
Early to mid-prophase cells that have been induced to return to interphase possess a normal interphase cytoplasmic microtubule complex. Prophase cells irradiated in the nucleus before this point return to interphase, whereas those irradiated after this point enter and complete mitosis with normal kinetics even if they contain broken chromosomes. To this end we show that the checkpoint responsible for this phenomenon becomes nonfunctional in PtK 1 cells ∼30 min before NEB. The goal of our study was to better characterize the radiation-induced reversion of prophase in vertebrate somatic cells, which has never been explored in a systematic manner. The inactivation of CDK1 then allows the cell to escape the mitotic condition and leads to the hallmark features of telophase including chromosome decondensation, nuclear envelope reformation, and cytokinesis. After this process has been initiated, another pathway is then triggered that inactivates the mitotic cyclin-dependent kinase (CDK1) by the ubiquination and subsequent proteolysis of its associated cyclin B (for reviews see 22, 45). Once all the kinetochores are attached and the wait anaphase signal is abrogated, APCs induce anaphase by degrading those proteins responsible for sister chromatid cohesion (e.g., 8, 12, 19, 54). 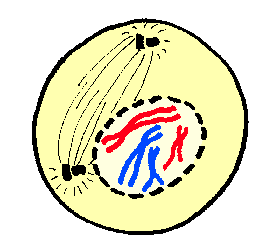
In part through the activity of Mad (e.g., 7, 37), Bub (e.g., 20, 56) and other gene products (like cdc20 21, 29), unattached kinetochores appear to inhibit the activity of ubiquitin ligases, known as anaphase-promoting complexes (APCs for review see 45), that target proteins for proteolysis. Within the past few years rapid progress has been made in understanding how this checkpoint works at the molecular level. This pathway is based on a negative feedback loop in which kinetochores release an inhibitor of anaphase until they are properly attached to spindle microtubules (Mts), and just one unattached kinetochore can delay anaphase for many hours. The checkpoint controlling the M–A transition monitors kinetochore attachment to the spindle (for review see 51). Together, these data reveal that a checkpoint control exists in early but not late prophase in vertebrate cells that, when triggered, reverses the cell cycle by apparently downregulating existing cyclin-dependent kinase (CDK1) activity. Immunofluorescent analyses reveal that the irradiation-induced reversion of prophase is correlated with the dephosphorylation of histone H1, histone H3, and the MPM2 epitopes. Thus, damage to an early prophase nucleus is converted into a signal that not only reverses the nuclear events of prophase, but this signal also enters the cytoplasm where it inhibits e.g., centrosome maturation and the formation of asters. If one nucleus in a cell containing two early prophase nuclei is selectively irradiated, both return to interphase, and prophase cells that have been induced to returned to interphase retain a normal cytoplasmic microtubule complex. Thus, the G 2 checkpoint that prevents entry into mitosis in response to nuclear damage ceases to function in late prophase. 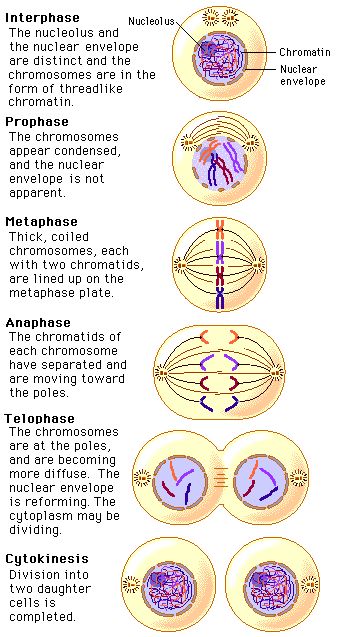
However, if early prophase nuclei are similarly irradiated, chromosome condensation is reversed and the cells return to interphase. When vertebrate somatic cells are selectively irradiated in the nucleus during late prophase (<30 min before nuclear envelope breakdown) they progress normally through mitosis even if they contain broken chromosomes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
